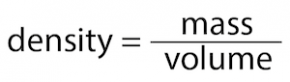Density, Temperature, and Salinity
Density
Density is a measure of how much mass there is in a given volume or amount of space. The density of any substance is calculated by dividing the mass of the matter by the volume of the matter.
|
|
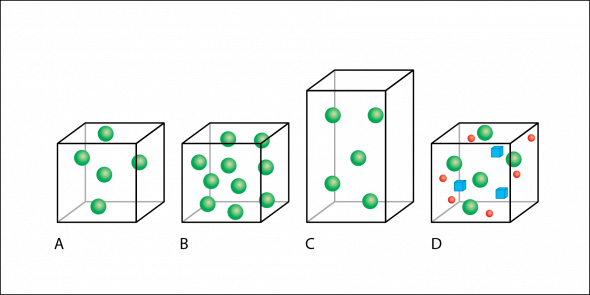
In Fig. 2.2, volume is represented by boxes and individual particles of matter are represented by colored shapes.
- Box A has five spheres.
- Box B is the same size, and has the same volume as box A, but box B has 10 spheres.
- Box C has the same mass as box A, with five spheres, but box C has a larger volume than boxes A and B.
- Box D has the same volume and number of green spheres as part A, but also includes other types of matter than the rest of the boxes—red circles and blue cubes.
Fig. 2.2. The boxes and colored shapes in this figure demonstrate the effects of changing mass and volume on density.
Image by Byron Inouye
If the amount of matter is increased without changing the volume, then the density increases (Fig. 2.2 A to 2.2 B). If volume increases without an increase in mass, then the density decreases (Fig. 2.2 A to 2.2 C). Adding additional matter to the same volume also increases density, even if the matter added is a different type of matter (Fig. 2.2 A to 2.2 D).
Salinity Affects Density
When salt is dissolved in fresh water, the density of the water increases because the mass of the water increases. This is represented by the addition of red spheres and blue cubes to the box from Fig. 2.2 A to Fig. 2.2 D. Salinity describes how much salt is dissolved in a sample of water. The more salt there is dissolved in the water, the greater its salinity. When comparing two samples of water with the same volume, the water sample with higher salinity will have greater mass, and it will therefore be more dense.
Temperature Affects Density
The density of water can also be affected by temperature. When the same amount of water is heated or cooled, its density changes. When the water is heated, it expands, increasing in volume. This is represented by the increase in the size of the box from Fig. 2.2 A to 2.2 C. The warmer the water, the more space it takes up, and the lower its density. When comparing two samples of water with the same salinity, or mass, the water sample with the higher temperature will have a greater volume, and it will therefore be less dense.
Relative Density
Fig. 2.3. (A) The bag filled with yellow liquid is floating on the surface. (B) The bag filled with orange liquid is floating in mid-water (subsurface floating). (C) The bag filled with green liquid has sunk to the bottom of the beaker.
Images by Byron Inouye
In Fig. 2.3, the beaker of liquid models a body of water like the ocean or a lake. The bag of liquid simulates a layer of water. The relative density of the liquid in the bag compared to the liquid in the beaker can be determined by observing whether the bag sinks or floats.
- In Fig. 2.3 A, the bag rose to the top of the beaker and is now floating on the surface. The yellow liquid and the bag are less dense than the liquid in the beaker.
- In Fig. 2.3 B, the bag is floating in mid-water (subsurface floating). The orange liquid and the bag are equal in density to the liquid in the beaker.
- In Fig. 2.3 C, the bag sank to the bottom of the beaker. The green liquid and the bag are more dense than the liquid in the beaker.
Water Layers
If water masses have salinity or temperature differences, they will form water layers because they have different densities. Water layers can sometimes be felt when swimming. For example, on hot days the sun’s heat can make water at the surface noticeably warmer than the deeper, cooler water. The relative density of one water mass in relation to another determines whether a layer of water floats or sinks.
Density and Buoyancy
Density can be determined by measuring the mass and volume of an object. In the Density Bags Activity, density was not calculated. Instead, relative density was determined by observing whether a bag of one liquid floated or sank in another liquid. A bag of liquid that sank was determined to be more dense than the liquid in the beaker. A bag of liquid that floated was determined to be less dense than the liquid in the beaker.
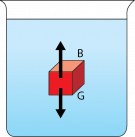
Fig. 2.5. Forces on a red block in water. The buoyant force is represented by the letter “B” and the upward pointing arrow. The gravitational force is represented by the letter “G” and the downward pointing arrow.
Image by Byron Inouye
The motion of any object is due to forces, which are pushes or pulls. Vertical—up-and-down—movement of water masses in the ocean can be explained in terms of two forces. The gravitational force (G) of the earth pulls downward and is proportional to the mass of an object. In Fig. 2.5, the gravitational force (G) is proportional to the mass of the red block. The gravitational force on an object is also called weight. The force due to gravity is greater on objects that are more massive, or weigh more. The buoyant force (B) of water pushes up. In the third century B.C., the Greek philosopher Archimedes was the first to describe buoyancy. He observed that the volume of water pushed out of a tub, or displaced, by an object was equal to the volume of the object. The buoyant force of the water is equal to the weight of the water displaced. This concept is known as Archimedes’ Principle, and it explains why objects sink or float. In Fig. 2.5, the buoyant force (B) is equal to the weight of the water displaced by the red block.
An object accelerates when the forces on that object are unequal. Although acceleration is commonly used to describe an object that is speeding up, the scientific definition of acceleration means changing speed. An accelerating object can be speeding up or slowing down. An object will always move in the direction of the greater force. An object may accelerate downwards (sink) or upwards (rise) in a body of water.
- Sinking is a downward vertical movement that occurs when the gravitational force (G) on an object is greater than the buoyant force (B) supporting it (G > B).
- Rising is the upward vertical movement that occurs when the gravitational force is less than the buoyant force (G < B).
If all of the forces on an object are balanced, there is no acceleration. In this case, the object may not move—like a book sitting on a flat table—or the object may move at a constant speed—like a car traveling at a steady 80 kilometers per hour. In the water, an object might remain still either at the surface or within the water column.
- Surface floating occurs when an object stays at the surface, because the forces are balanced at the surface (G = B).
- Subsurface floating, or neutral buoyancy, occurs when an object maintains its position in mid-water, neither sinking nor rising (G = B).
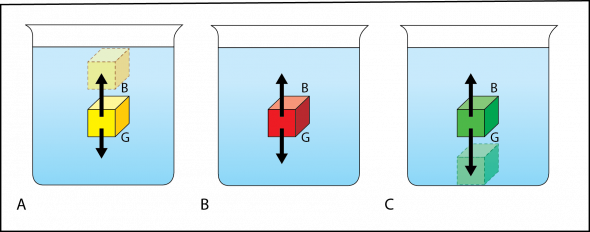
Fig. 2.6. The yellow cube (A) is less dense than water, the red cube (B) has the same density as water, and the green cube (C) is more dense than water. The buoyant force is represented by the letter “B” and upward pointing arrows. The gravitational force is represented by the letter “G” and downward pointing arrows.
Images by Byron Inouye
Three cubes of the same size, but with different masses and thus different densities, are placed in three beakers of water (Fig. 2.6). Because the cubes are identical in volume, they displace the same amount of water. By Archimedes’ Principle, the buoyant force (B) acting on each cube is equal. Buoyant force is represented in Fig. 2.6 as upward pointing arrows, indicating the water is pushing up on the cubes. These arrows are the same length for each of the cubes, indicating that the strength of the buoyant force acting on each cube is the same.
Because the masses of the cubes are not equal, the gravitational force (G) acting on each cube is different. Gravitational force is represented in Fig. 2.6 as downward pointing arrows, indicating the gravitational force is pulling down on the cubes. These arrows are different lengths for each cube, indicating that the amount of the gravitational force is different for each cube. The downward pointing arrow in Fig. 2.6 A is the shortest, indicating that the yellow cube has the least mass and is the least dense. The downward pointing arrow is the longest in Fig 2.6 C, indicating that the green cube has the most mass and is the most dense.
The density of the cube relative to the density of water determines if the cube will float, sink, or be neutrally buoyant:
- If the density of the cube is less than the density of the water, gravitational force will be less than the buoyant force (G < B), and the object will rise to the surface (Fig. 2.6 A).
- If the density of the cube is equal to the density of the water, the cube will float in middle of the column of water because the gravitational force and buoyant force are balanced (G = B). This cube is neutrally buoyant (Fig. 2.6 B).
- If the density of the cube is greater than the density of the water, the cube will sink because the gravitational force is greater than the water’s buoyant force (G > B) (Fig. 2.6 C).