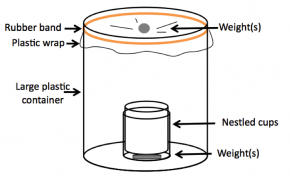
Fig. 5-21: Completed food-safe still.
Title
Activity: Simulate the Water Cycle
Table of Contents
In this activity we will simulate the water cycle. The apparatus we will be using is called a still. A still is a closed system. A closed system allows energy, such as sunlight or heat, but not matter, to cross its boundaries. An open system, on the other hand, allows both energy and matter to cross its boundaries.
Materials
A. For food-safe stills:
- Salt water made in a food-safe container, like a pitcher
- Fresh water stored in a food-safe container, like a pitcher
- Food coloring
- Ruler
- Scissors
- Two large, tall, food-safe plastic containers
- Four small food-safe plastic cups
- At least four flat weights (e.g. washers or coins)
- Two sheets of plastic wrap
- Two rubber bands
- Food-safe spoons
B. For non food-safe stills:
- Salt water
- Fresh water
- Food coloring
- Two 2000 mL beakers
- Two 100 mL beakers
- Two 250 mL beakers
- Two thermometers
- Two small pieces of tubing
- Two sheets of plastic wrap
- Two rubber bands
- Two small weights (e.g. small stones)
- Two hot plates (or heat lamps)
- Graduated cylinder
- Four evaporating dishes (e.g. Petri dishes, optional)
- Dissecting microscope (optional)
- Conductivity apparatus (from Conductivity Activity, optional)
Procedure
A. For food-safe stills:
- Color the salt and fresh water with equal amounts of food coloring.
- If the cups are more than half as tall as the large plastic container, cut them until they are shorter (see Fig. 5-21).
- In the bottom of two cups, place flat weights. Place a second cup inside of the first (see Fig. 5-21).
- Place the nestled cups into two clean, large food-safe containers. Fill the bottom of one of the tall plastic containers with ~3 cm of fresh water, and the bottom of the second with ~3 cm of salt water.
- If the large containers are the same, the amount of the water in each should be the same.
- If the containers are different, the amount of water will vary based on the size of your container.
- If the cups are floating, remove some water or add more weights until they are stable.
- Label each still as FRESH or SALT.
- Loosely cover the tops of the tall containers with plastic wrap. Secure the plastic wrap with a rubber band (see Fig. 5-21).
- Place a small weight in the center of each of the plastic wrap squares so that a cone-shaped depression forms. Make sure the bottom of the depression is directly over the cups and that the plastic wrap is not touching the beaker. The completed still should look like Figure 5-21.
- Put the stills outside in a sunny location. To make sure the stills do not fall over, prop them up with rocks or other materials.
- After at least 24 hours, collect your stills. Record your observations in Table 5.1.
- Predict how the liquid inside the cups will taste compared to the liquid remaining in the large plastic containers. Record your predictions in Table 5.1. Test your prediction by tasting the water with spoons. Record your observations in Table 5.1.
- If you have additional water in the cups after tasting, further test the liquid using one of the methods described in Section B, Procedure 10.
B. For non food-safe stills:
- Color the salt and fresh water with equal amounts of food coloring.
- In a 2000 mL beaker, place 150 mL of fresh water. In the second 2000 mL beaker, place 150 mL of salt water. Label each beaker.
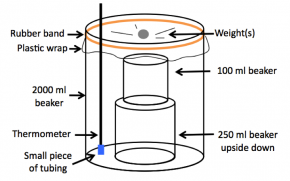
- Place an upside down 250 mL beaker in the middle of each larger beaker. As you place this beaker at the bottom, tip it upward so that water flows under it. Place a 100 mL beaker on top of each 250 mL beaker (see Fig. 5-22).
Fig. 5-22: Completed non-food-safe still.
- Loosely cover the tops of the large beakers with plastic wrap. Secure the plastic wrap with a rubber band (see Fig. 5-22).
- Place a small weight in the center of each of the plastic wrap squares so that cone-shaped depressions form. Make sure the bottom of the depression is directly over the small beaker and that the plastic wrap is not touching the beaker.
- Place a small piece of tubing over each of the bulbs of two thermometers. Poke one thermometer through the plastic wrap of each still through to the water at the bottom of the large beaker. The completed still should look like Figure 5-22.
- Place the stills on hot plates or under heat lamps. If using a hot plate, monitor the still to make sure the water does not exceed 90ЛљC.
- At the end of the experiment, turn off the hot plates or heat lamps. Record your observations about the still in Table 5.2.
- Determine the total amount of liquid collected in the small beakers of each of your stills by measuring the liquid in a graduated cylinder. Record your measurements in Table 5.2.
- Test the liquids you collected using one or more of the following methods:
- a. Evaporation
- Predict how the liquid inside the cups or small beakers will appear compared to the liquid remaining the in large containers once evaporated.
- Test your prediction by putting equal amounts of liquid into small evaporating dishes. Bake or place the dishes in the sun until all the liquid is gone.
- Look at each sample under a dissecting microscope.
- Record your observations.
- Conductivity
- Predict how conductive the liquid inside the cups or small beakers will be compared to the liquid remaining in the large containers.
- Test your prediction by using the conductivity apparatus from Unit 3 Properties of Water - Conductivity Activity.
- Record your observations.
- a. Evaporation
Activity Questions
- Compare the results from the fresh water and salt water stills. How are they similar? How are they different?
- Explain how a still works in terms of evaporation and condensation (see Fig. 5.20).
- Distilled water is water from which solids and dissolved particles have been removed. How does a still remove solids and dissolved particles?
- If you made a food-safe still: What sources of error may affect affected your results? Why?
- If you made a non-food-safe still: Determine the condensation rate of the stills by dividing the total mL in the small beakers by the number of hours the still was operating.
- What was the condensation rate of the fresh water still?
- What was the condensation rate of the salt water still?
- Compare your still to the water cycle. How is a still similar to the water cycle? How is it different?
- What could be done to make stills produce more water in the same period of time?
- What techniques might be used to
- purify water?
- obtain fresh water from salt water?
- obtain fresh water from moist soil?
- You are lost in the desert. Explain how you might use your knowledge of still construction to survive. Include a list of the minimum materials you would need to carry with you and those you would need find in the environment.
- Label the following systems as closed or open. Explain your reasoning.
- Earth
- The ocean
- An aquarium
- A terrarium–a big glass jar with plants that has a tightly sealed lid