Heterotrophs
Energy acquisition is important for all living organisms to grow, reproduce, and move. Heterotrophs are organisms that cannot produce their own food. Heterotrophs must consume other organisms in order to acquire energy.
Humans are heterotrophs, and eating food has united people across cultures for thousands of years. Food is important in cultural rituals and celebrations, from birthday cake to holiday meals. And yet, the ultimate purpose of eating food is to provide the body with usable energy—whether the meal is a fancy feast, an everyday meal, or an on-the-go snack.
Autotrophs
Autotrophs, by contrast, are living organisms capable of producing their own food. The term autotroph comes from two Greek root words. Auto means self, and troph means to feed.
Autotrophs fall into two broad categories: chemo-autotrophs and photo-autotrophs. Chemo-autotrophs are autotrophs capable of producing organic (carbon-containing) compounds without the sun’s energy. Chemo-autotrophs use carbon dioxide (CO2) as their carbon source. They get energy from the chemical bonds in inorganic (non-carbon) compounds like hydrogen sulfide, elemental sulfur, ferrous iron, molecular hydrogen, and ammonia.
Fig. 2.39. Riftia pachypitila is a large tubeworm. It has a symbiotic relationship with a chemoautotrophic bacterium that oxidizes hydrogen sulfide from hydrothermal vents.
Image courtesy of National Oceanic and Atmospheric Administration (NOAA) Ocean Explorer
Most chemo-autotrophs are bacteria or archaea. They tend to live in hostile environments, such as deep sea vents (Fig. 2.39). In these ecosystems, chemo-autotrophs are often the main primary producers. Primary producers are autotrophs that produce food (sugars) from inorganic compounds (like carbon dioxide). Most other autotrophs are photosynthesizers, like plants and algae, which use energy from the sun.
Photo-autotrophs are autotrophs that get their energy from sunlight. Familiar examples of photo-autotrophic organisms are algae and vascular plants. Photo-autotrophs use the energy in sunlight to make food through photosynthesis.
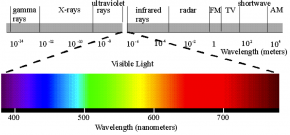
Fig. 2.40. This diagram of the electromagnetic spectrum emphasizes the small portion of the spectrum that is visible to human eyes. Wavelengths are measured in meters (m) along the grey bar and in nanometers (nm) along the colored bar showing visible light.
Sunlight is actually a form of electromagnetic radiation. Electromagnetic radiation has wave-like behavior with both electric and magnetic components. The electromagnetic spectrum shows the range of electromagnetic radiation (Fig. 2.40). Gamma rays have the shortest wavelength and highest frequency. Radio waves have the longest wavelength and lowest frequency. The majority of solar radiation that reaches earth’s surface is in the visible range of the electromagnetic spectrum.
Sunlight is made of white light. White light contains all of the colors of the visible spectrum. White light can be separated into its component colors by prisms or water droplets, resulting in rainbows. The wide range of colors on earth is possible because the sun emits the entire range of visible light wavelengths.
We see color when an object absorbs, reflects, or transmits light. When white light hits a solid object, like a red stop sign, all colors except red are absorbed. The wavelengths that are reflected are the ones perceived by the human eye as colors. Red wavelengths are not absorbed by the stop sign; they are reflected, or bounce, off the sign. People see the stop sign as red because they see the reflected red light.
Objects that are translucent allow light to pass through them. For example, when light enters blue glass, only blue wavelengths pass through, or are transmitted. People see the glass as blue because the glass absorbs all of the visible wavelengths except blue.
Most of the colors we see are not due to the reflection or transmission of a single wavelength, but rather a range of wavelengths. For example, a green plant may reflect a range of blue, green, and yellow wavelengths. This helps to explain why there are so many different shades of green.
Pigments
Plants use pigments to capture light energy to power photosynthesis. Pigments are colored compounds that absorb specific wavelengths of light. Chlorophyll is the primary pigment in photosynthesizing plants. There are three forms of chlorophyll pigment: chlorophyll a, chlorophyll b, and chlorophyll c. Each form of chlorophyll absorbs different wavelengths of light that can be used for photosynthesis.
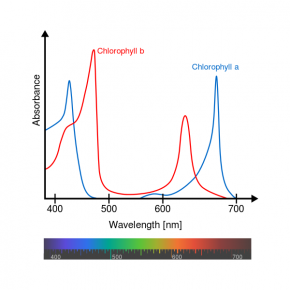
Fig. 2.41. Relative absorbance of the visible wavelengths in sunlight by the pigments chlorophyll a and chlorophyll b
An absorption spectrum is a graph that shows how pigments absorb different wavelengths of light. The primary photosynthetic pigments in green algae and vascular plants are chlorophyll a and chlorophyll b. The absorption spectrum for chlorophyll a and chlorophyll b (Fig. 2.41) shows how well each pigment absorbs wavelengths in the visible light section of the electromagnetic spectrum.
The absorption spectrum shows that chlorophyll a and chlorophyll b have maximum absorbance in the blue and red ends of the spectrum. There is almost no absorbance in the green range. The reason green algae and most vascular plants appear green is that green light is reflected back to our eyes while other wavelengths are absorbed.
Many algae and vascular plants have other pigments in addition to chlorphyll. These other pigments absorb light at different parts of the spectrum. For this reason, algae and vascular plants vary widely in their absorption spectra and also in the colors that they appear to be. Table 2.7 shows the primary pigments associated with the major algae groups.
Table 2.7. Characteristic pigments of the main algae groups*
| Group |
Chlorophyll Pigment |
Other Pigments |
| Blue-green algae |
chlorophyll a
(some also have chlorphyll b) |
phycobilins and various carotenoids |
| Green algae and vascular plants |
chlorophyll a and b |
various carotenoids |
| Red algae |
chlorophyll a |
phycobilins |
| Brown algae |
chlorophyll a and c |
carotenoids (beta-carotene, fucoxanthin, and violaxanthin) |
| Diatoms |
chlorophyll a and c |
fucoxanthin |
* Source: Huisman, Abbott, and Smith. 2007. Hawaiian Reef Plants. University of Hawaii Sea Grant College Program. Honolulu, HI.
Carotenoid pigments have a brown, yellow, or red color. They are present in blue-green, green, and brown algae. The carotenoids pigments include compounds called fucoxanthins, peridinin, and beta-carotenes.
Phycobilins are another set of pigments. These include phycoerythrins, which are red pigments, and phycocyanobilins, which are blue.
Activity
Investigate the effect of wavelength on the rate of photosynthesis using the aquatic plant Elodea.